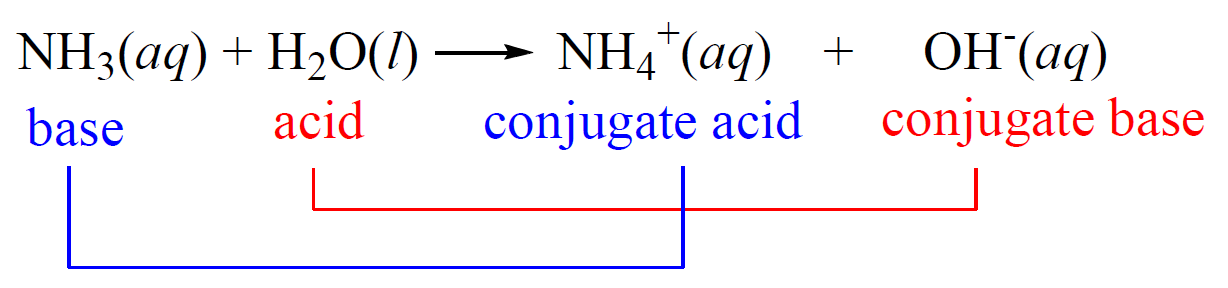
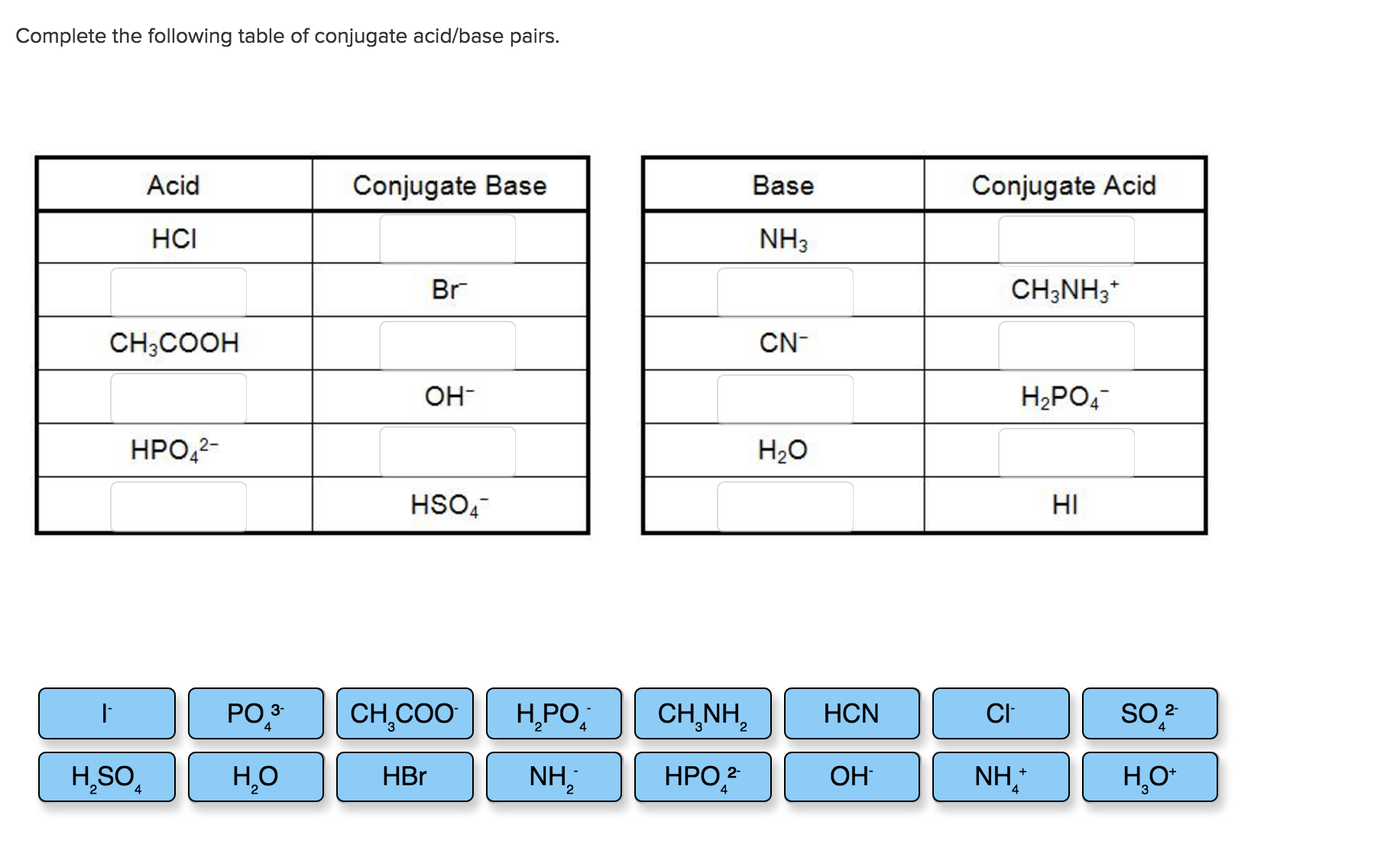
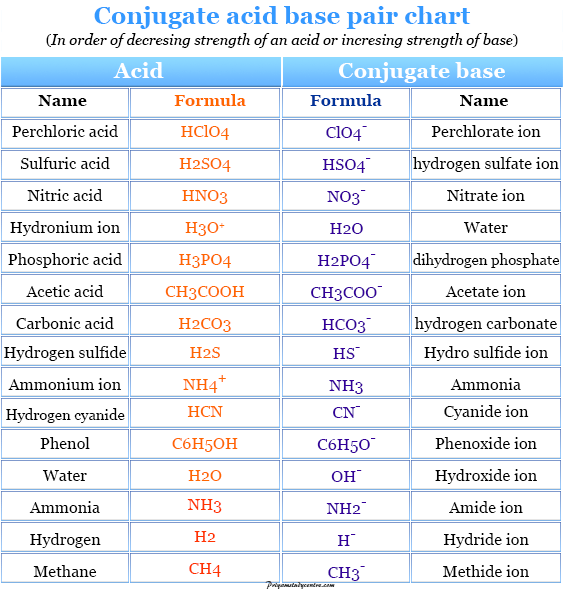
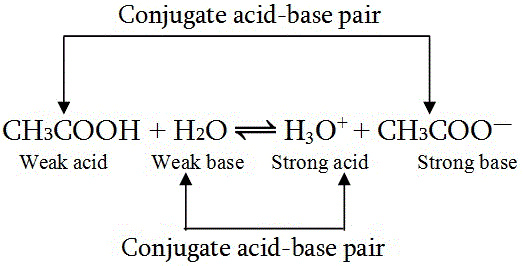
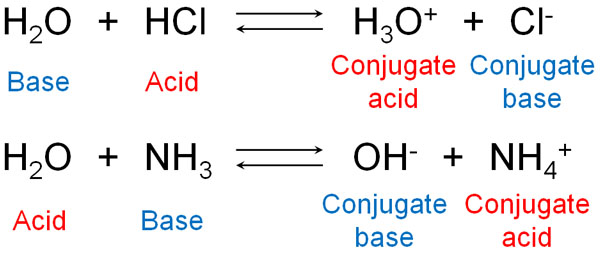
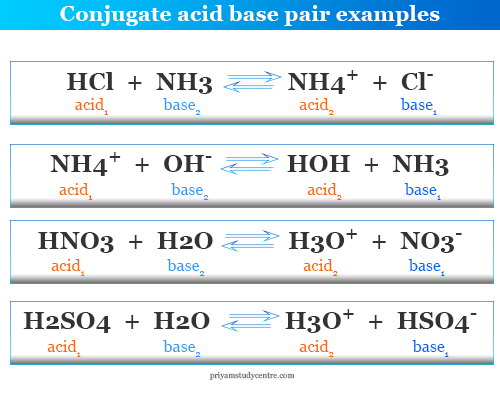
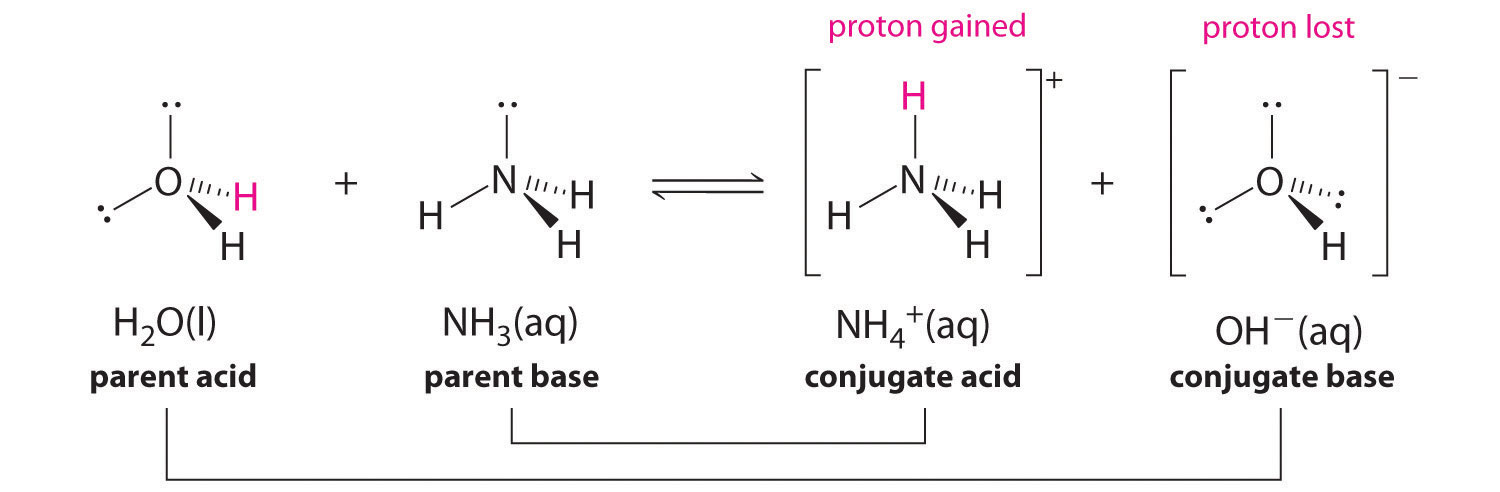
How to Identify Acid, Base, Conjugate Acid, and Conjugate Base Examples and Practice Problems - YouTube
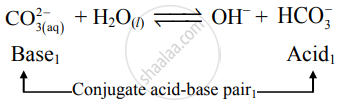
Label the one conjugate acid-base pair in the following reaction. COX3(aq)2−+HX2OX(l)↽−−⇀OHX−+HCOX3− - Chemistry | Shaalaa.com
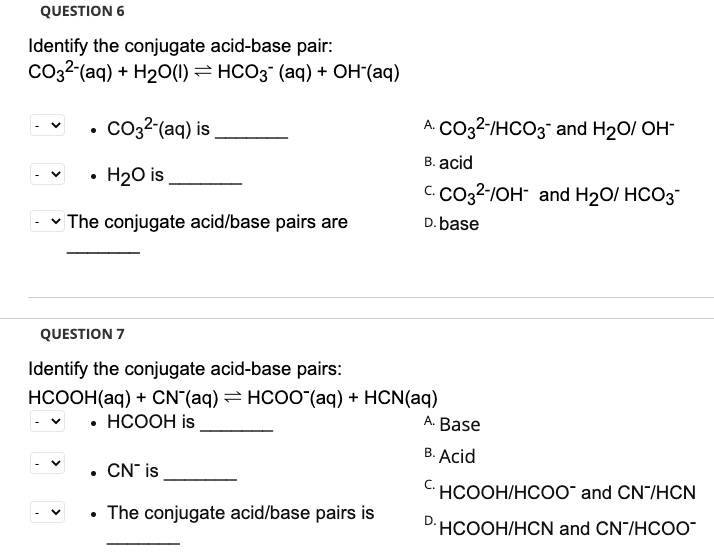
SOLVED: QUESTION 6 Identify the conjugate acid-base pair: CO32-(aq) H2O() = HCO3" (aq) + OH(aq) CO32-(aq) is CO32-IHCO3" and H2O/ OH" acid CO32-IOH- and H2O/ HCO3 base H2O is The conjugate acidlbase

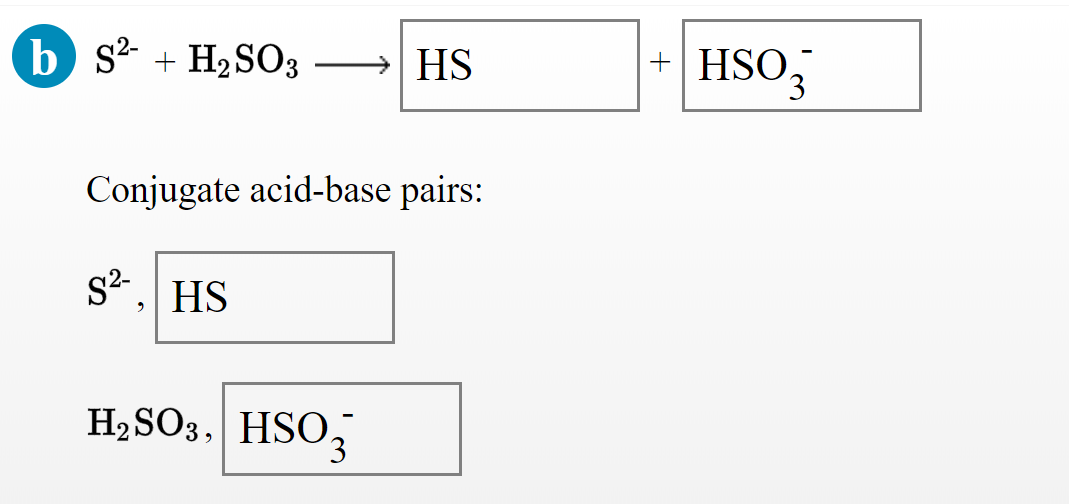
What is meant by the conjugate acid-base pair? Find the conjugate acid/ base for the following species: HNO 2 , CN - , HClO 4 , F - , OH - , CO 3 2- and S 2-