
Identify the conjugate acid-base pairs in the following reaction. Indicate what each substance is in each pair. H2CO3 + PO43- arrow HCO3- + HPO42- | Homework.Study.com

SOLVED: Konsider the reaction: H2SO3 HCO3" H2CO3 HSO3" a) Identify the acid, base, conjugate acid and conjugate base b. What is the conjugate base of water? C What is the value of [
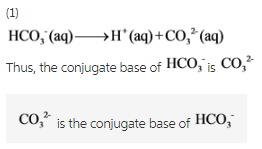




![pHe/ [HCO3-] (Davenport) diagram of the extracellular acid–base... | Download Scientific Diagram pHe/ [HCO3-] (Davenport) diagram of the extracellular acid–base... | Download Scientific Diagram](https://www.researchgate.net/publication/232279532/figure/fig5/AS:216381174882304@1428600700010/pHe-HCO3-Davenport-diagram-of-the-extracellular-acid-base-variables-Acid-base.png)



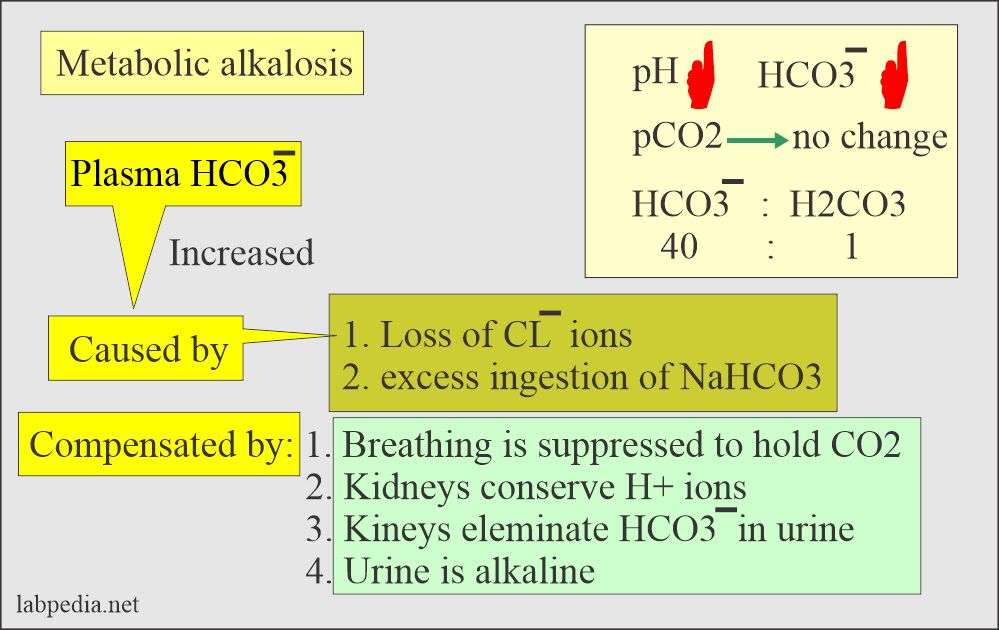
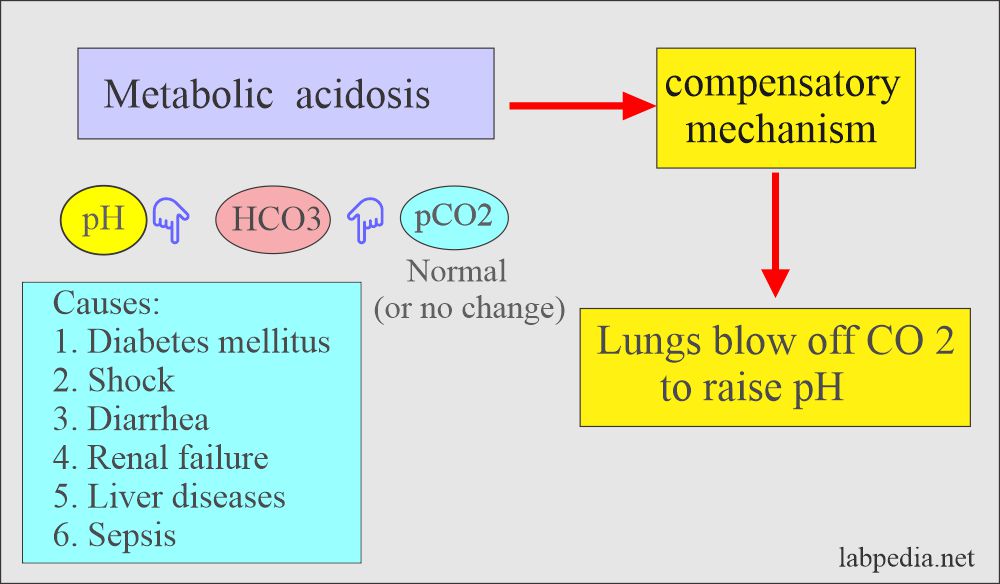



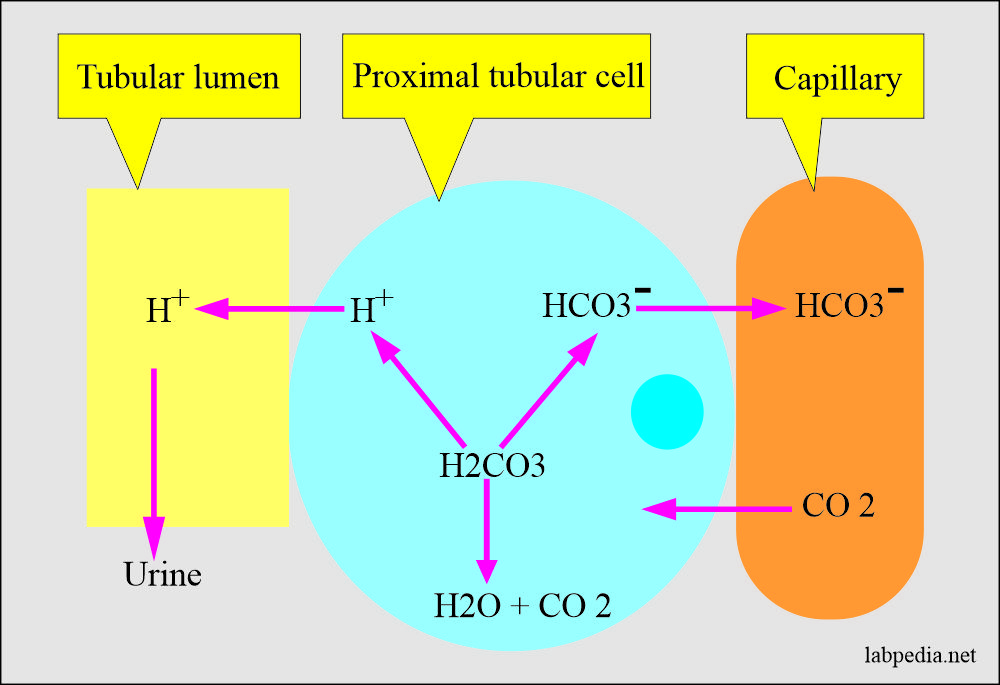
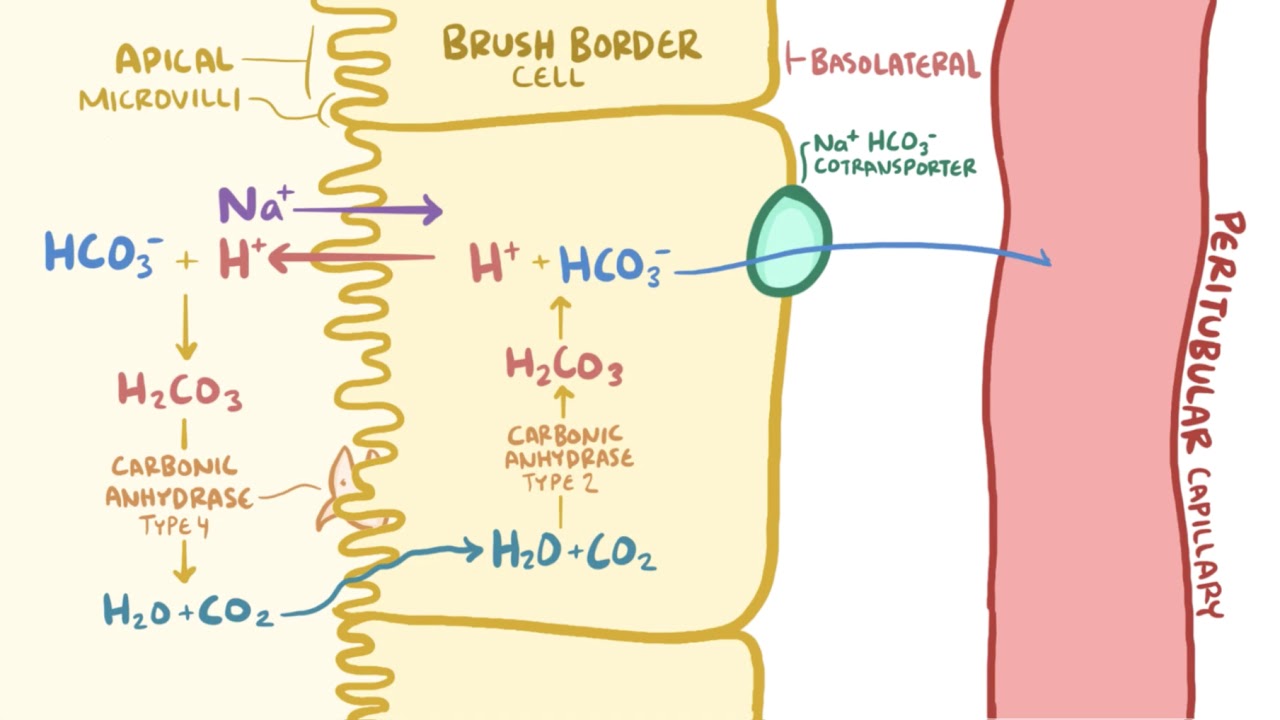
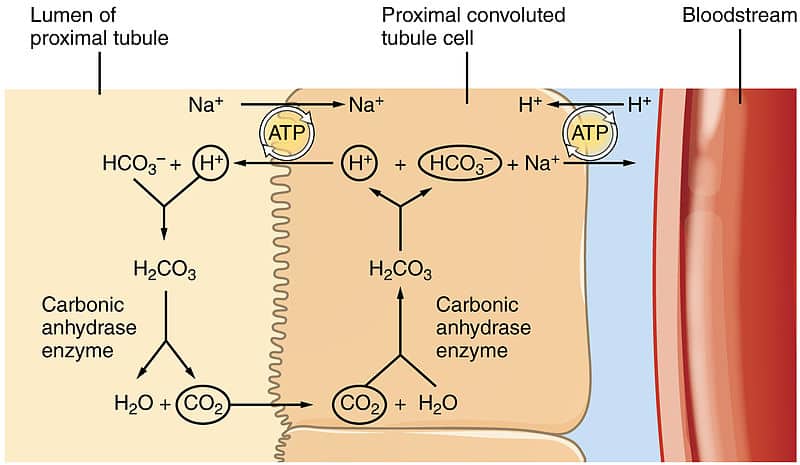
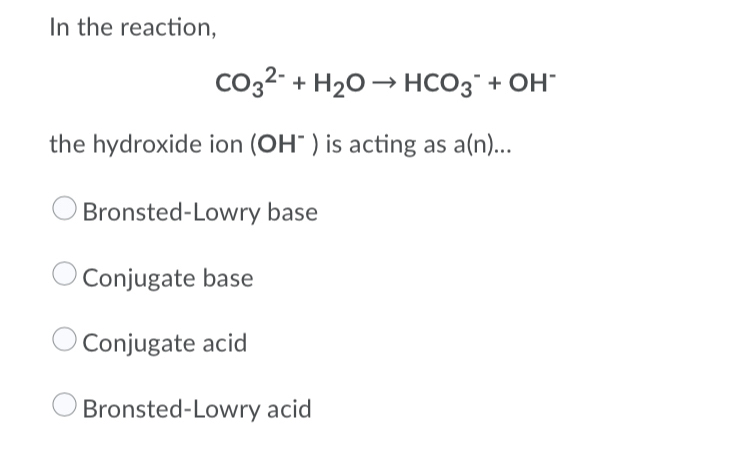



![ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/53160915-1659276540.7997923.jpeg)
