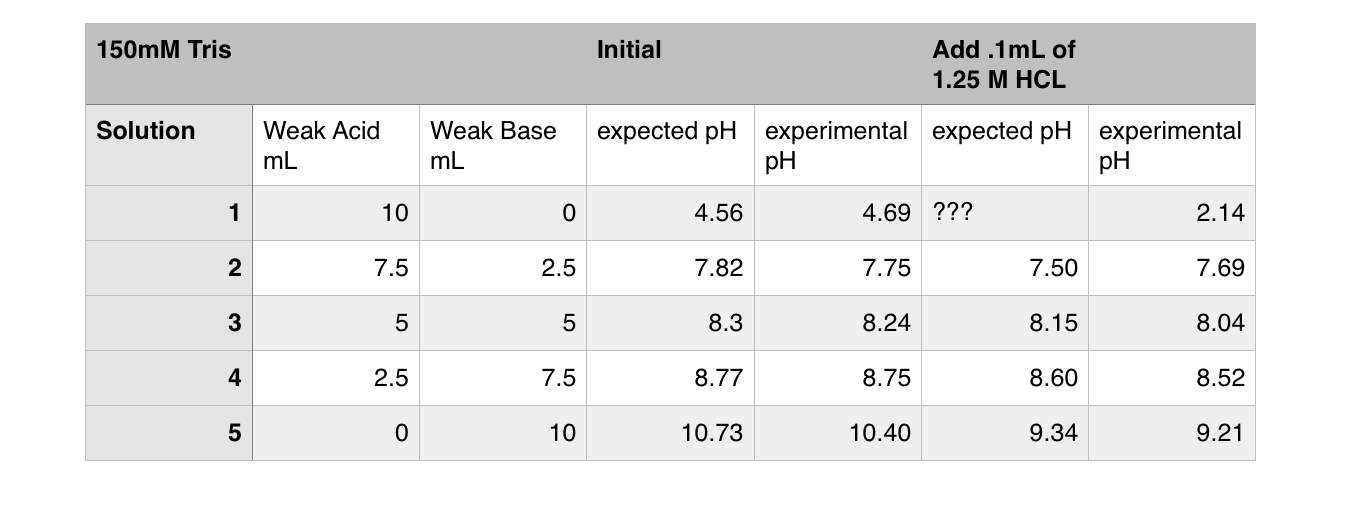
WO2014165660A1 - Pharmaceutical formulations for subcutaneous administration of furosemide - Google Patents

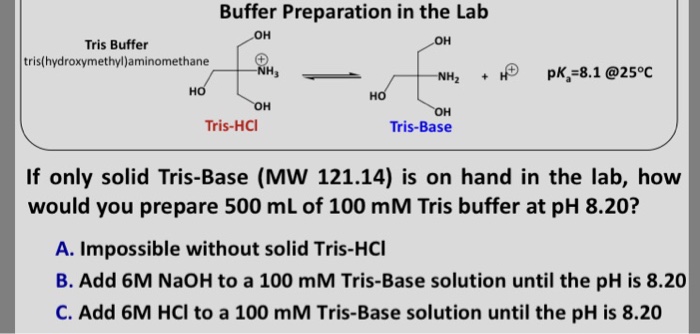
Tris(hidroximetil)-aminometano TRIS base is useful in the pH range of 7.0-9.0. Has a pKa of 8.1 at 25°C. | Sigma-Aldrich

SOLVED: Tris' is a buffer often used in biochemistry. We can simplify the equilibrium reaction as: Tris-HCl + H2O -> Cl- + H3O+ + Tris with pKa = 8.072 and the molar