Sub-microscopic representation: (a) strong acid 0,01M (b) Weak acid... | Download Scientific Diagram

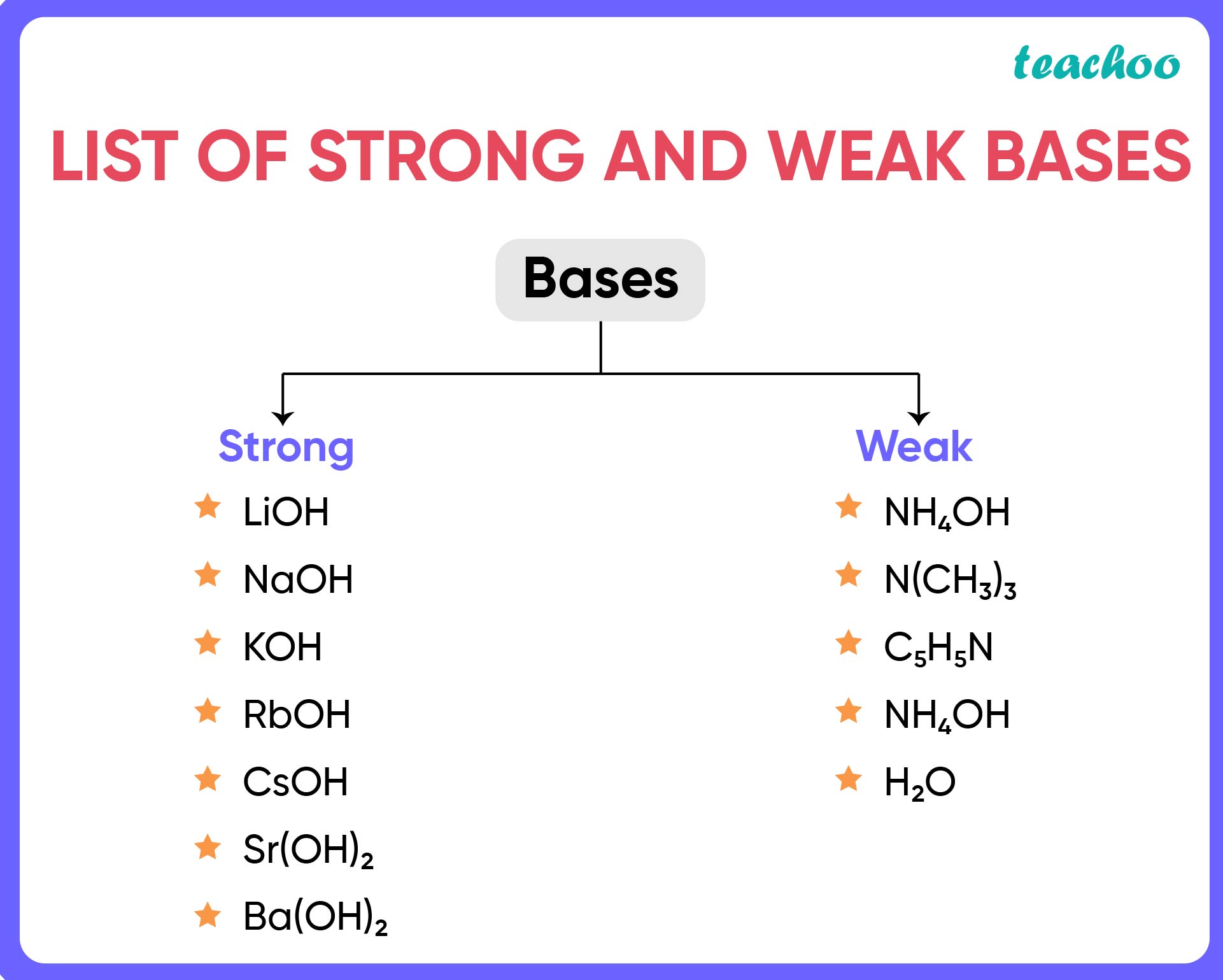
What is meant by strong bases and weak bases? Classify the following into strong bass and weak - YouTube

SOLVED: Which of the following ions will act as a weak base in water? a) CIO- b) NO3- c) Cl- d) Br- e) None of the above will act as a weak

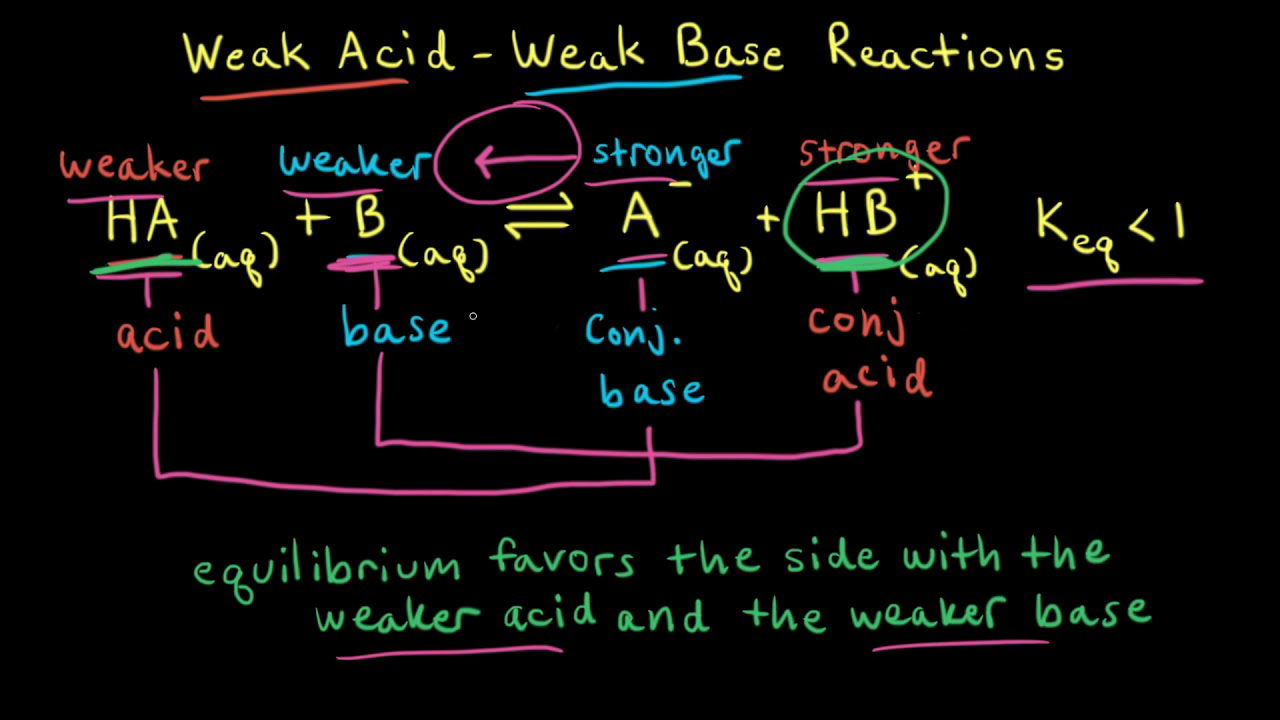
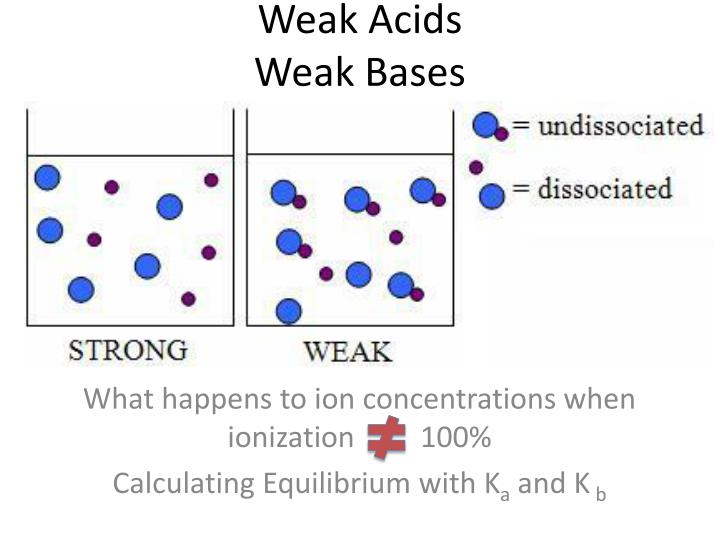
Unit 12 Weak Acids and Weak Bases. What makes a Weak Acid? Weak acids (covalently bonded H compounds) are ionized by water less than 100% HBrO + H 2 O. - ppt download
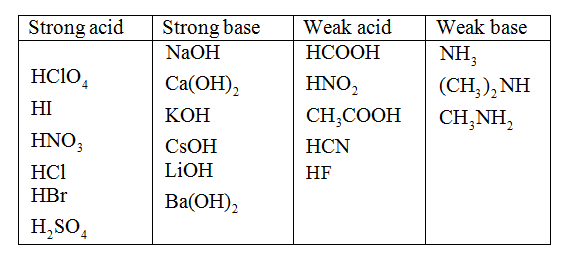
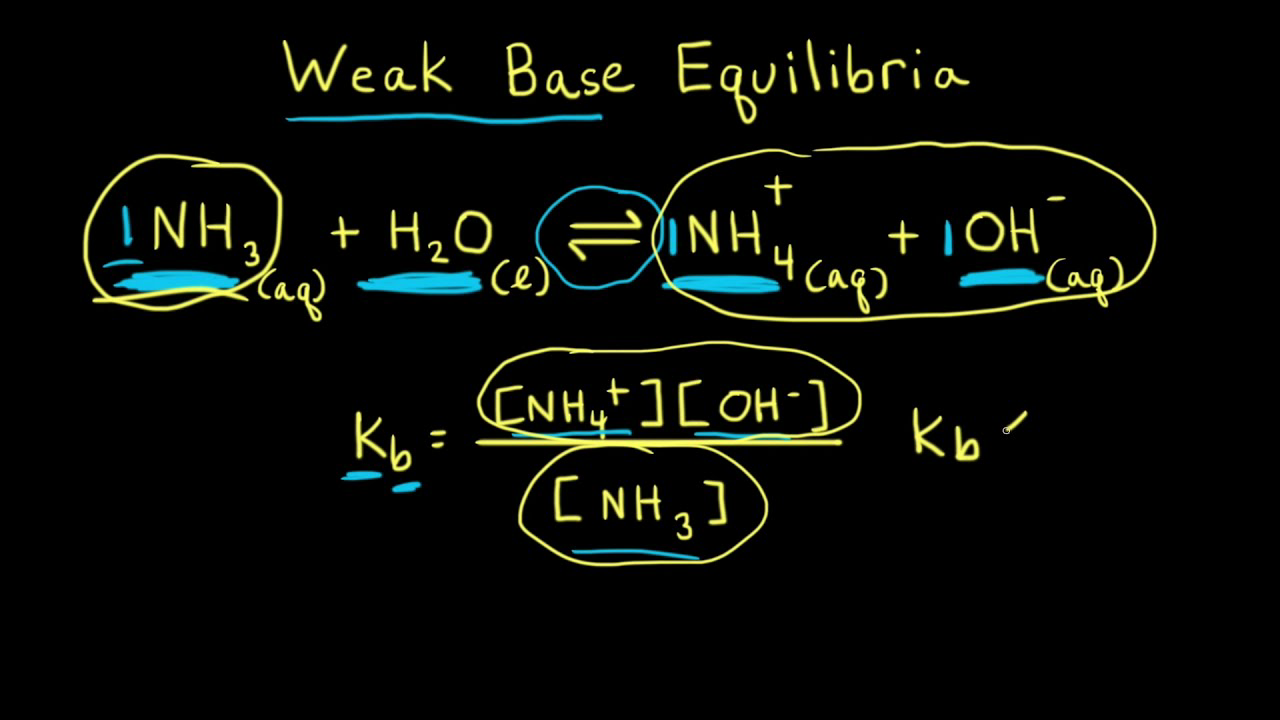



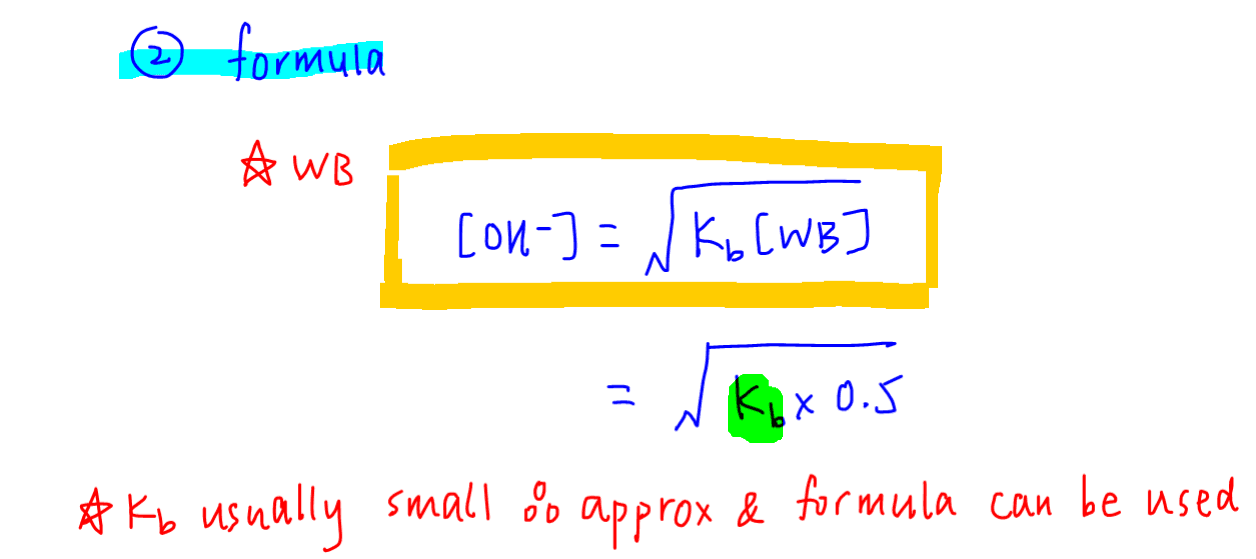

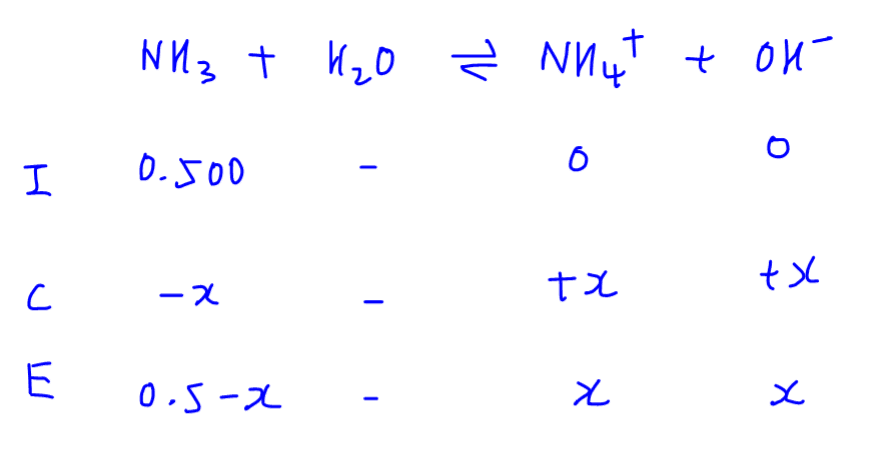


![Difference between Strong and Weak Base - with Examples [in Table] Difference between Strong and Weak Base - with Examples [in Table]](https://d77da31580fbc8944c00-52b01ccbcfe56047120eec75d9cb2cbd.ssl.cf6.rackcdn.com/0e323ff3-079c-4d5e-b21f-0f4c25082521/differences-between-strong-and-weak-bases-01.jpg)